The Center’s research focuses on developing innovative nanotechnology platforms and biomaterials for drug delivery and regenerative medicine which include:
-
- Biomaterials functionalization to target immune system to enhance regeneration
- Synthesis and functionalization of biomimetic nanoparticles to target and treat inflammatory-based conditions
- Biomimetic tridimensional in vitro system to study complex cancer environment
- Drug delivery system for the sustained release of proteins, cytokines, antibiotics and analgesics
- Smart materials development (injectable and implantable) to enhance surgery outcome
The rate at which nanotechnology and tissue engineering has seen innovations move from bench to bedside has been slower than expected. The demand for products with desired properties for clinical therapy (high targeting, low toxicity) and surgical applications (high biocompatibility, compliance with organ function) remains elevated. We recognized that the body has incredible healing capabilities and have worked to harness these to their fullest potential by creating biomimetic biomaterials, injectable or implantable, able to elicit desired cells and tissue responses.
The Center for Musculoskeletal Regeneration is focused on the development of cutting edge nano- and bio-materials for therapy. Our goal is to develop biomaterials which we either use as in vitro model of complex system, injectable nanotherapies or implantable synthetic tissues.
Our efforts are directed towards the treatment of currently unmet traumatic and pathological conditions. By combining cell biology with nanotechnology, we create innovative medical technologies that work within, and not around, the laws of nature.
Biomimetic injectables
We developed a new class of biomimetic nanoparticles inspired by the ability of leukocytes to target inflammation and infiltrate inflamed tissues. By using the cell membrane proteins of different immune cells as building blocks, we created injectable nanoparticles able to avoid reticuloendothelial clearance, specifically target inflamed endothelium, cross the endothelial barrier, and increase accumulation of therapeutic payloads in target tissue.
The strategy of targeting inflammation, allowed us to use our biomimetic nanoparticle for the treatment of different inflammatory-based pathologies that affect humans including cancer, trauma, and infection. We are committed to focus their application on open orthopedic applications such as osteoporosis, osteoarthritis, bone metastasis and osteosarcoma.
Moreover, our platform is very versatile on its capacity of loading different molecules, ranging from small drugs, protein, cytokines, and nucleic acid such as miRNA and siRNA.
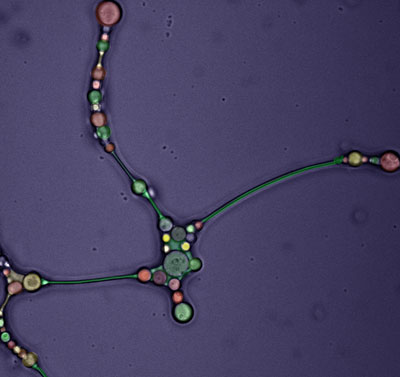
Biomimetic Implantables
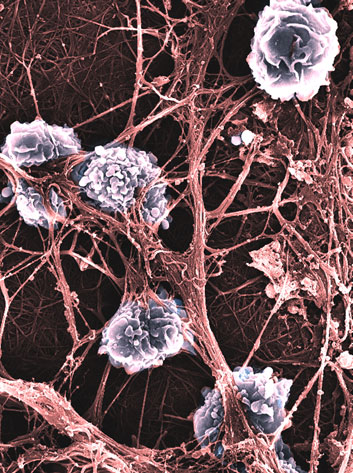
Our approach to biomaterials for tissue engineering is focused on studying the reactions of inflammatory cells such as macrophages, dendritic, and T cells, to biomaterials. We demonstrated that all regenerative processes are dependent on a complex dialogue between multiple cell types, also involving the chemical and physical cues provided by the surrounding microenvironment. We functionalize our materials with sugar moieties which allow for the activation/polarization of different immune cells.
Macrophages are key players in this response. The activation of M1 macrophages leads to adverse immune dysfunction, fibrosis, and pathological complications. Conversely, M2 switching is associated with faster tissue healing and can prevent scar tissue formation. Through our studies, we showed that immune and stem cells respond to an implanted material according to its composition, structure, and surface properties.
To augment implant biocompatibility and minimize immunogenic response, we developed several immune-instructive biomaterials able to tune the immune-response towards a faster and improved regeneration.
Tridimensional tissue model
It is well recognized that one of the major drawbacks of using traditional two dimensional cultures to model the living systems is inaccurately reflecting the physiological manner in which modulators, nutrients, oxygen, and metabolites are applied and removed.
We exploit our ability to develop biomimetic materials to create in vitro models that resemble the features of physiological or diseased tissues using different type of fabrication: electrospinning, 3D bioplotter, microfluidic devices.
We are able to mimic complex tissues varying composition, mechanical features, biological factors and using different cell types. We are particularly focused on the role of extracellular matrix in homeostasis and disease and epigenetic.
Our tridimensional systems used with human primary cell lines are the models that most likely resemble the human scenario.

Translational Biomaterials for Unmet Surgical Needs
Our group is actively involved to develop translational biomaterials with the help and support of HMRI cGMP for surgical use. We have applied our expertise in biomaterials to developed different type of devices for the local and sustained release of antibiotics, small drug and NSAID.
We are particularly focus on keeping an open conversation with our clinical counterpart to develop technologies which could improve the clinical practice.
We normally ask to our clinicians: what do you need in the OR and you don’t have it?
As a testimony to our translational research, we are also conducting the first large-scale FDA preclinical study to test the osteoregenerative capability of a novel biocompatible device for bone healing.
